
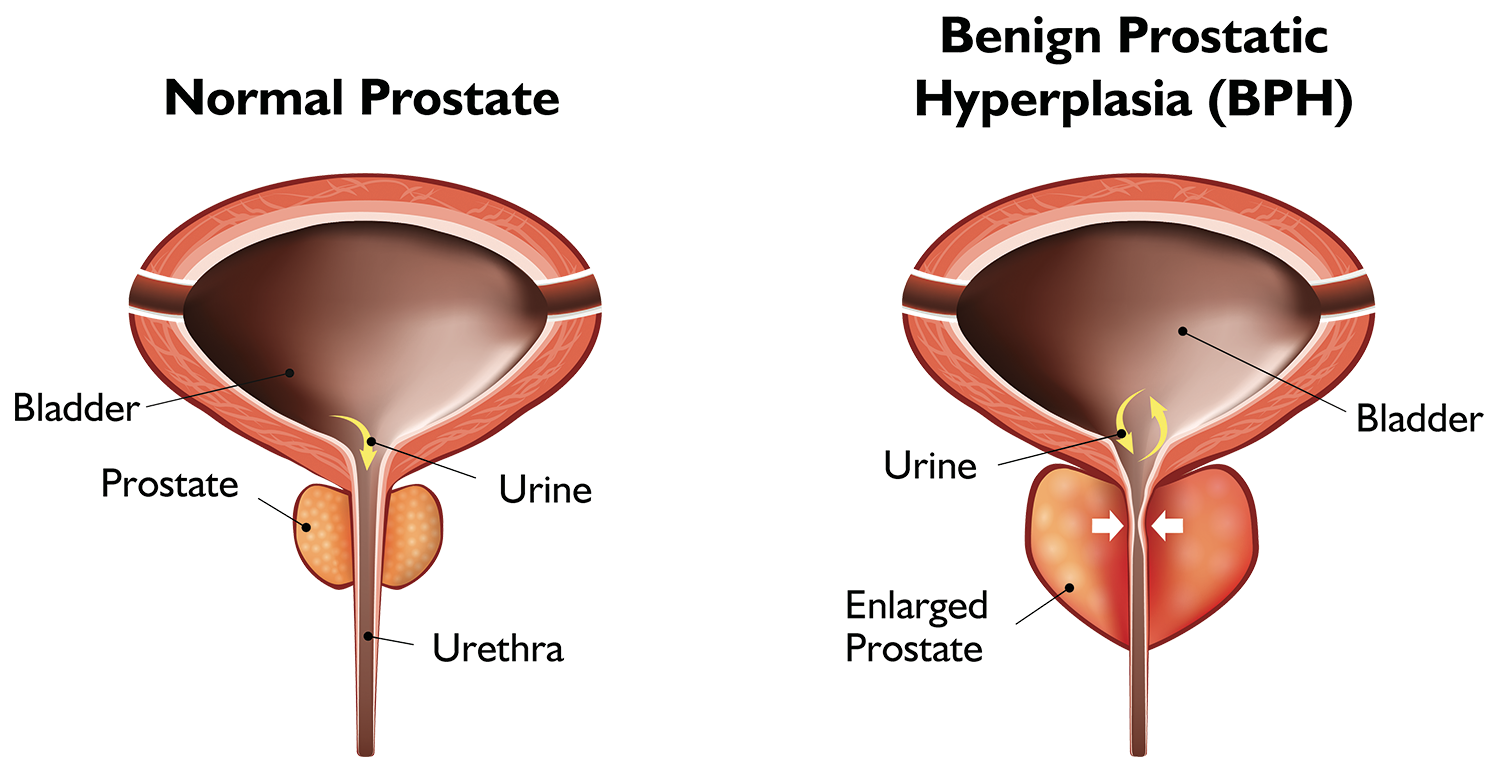
What is BPH?
Benign Prostatic Hyperplasia, or BPH, is a condition in which the prostate enlarges as men get older. BPH is a very common condition that affects over 42 million men in the US1 and over 660 million aging men worldwide. Over 40% of men in their 50s and over 70% of men in their 60s have BPH. While BPH is a benign condition and unrelated to prostate cancer, it can greatly affect a man’s quality of life.
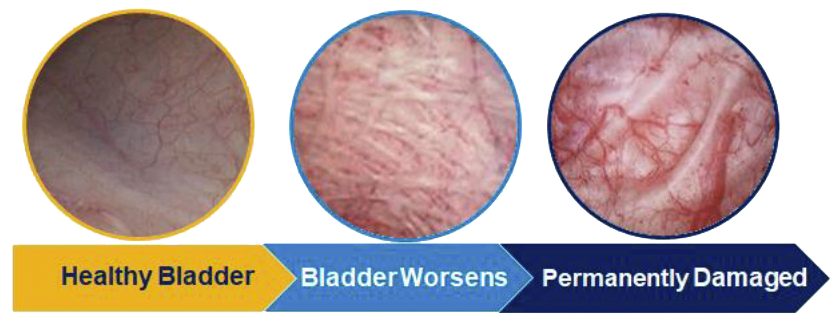
As the prostate enlarges, it presses on and blocks the urethra, causing bothersome urinary symptoms. If left untreated, BPH can lead to permanent bladder damage.
Symptoms include:
- Frequent need to urinate both day and night
- Weak or slow urinary stream
- A sense that you cannot completely empty your bladder
- Difficulty or delay in starting urination
- Urgent feeling of needing to urinate
- A urinary stream that stops and starts
Symptoms of BPH can cause loss of productivity, depression, and decreased quality of life.
If you suffer from the above symptoms, you are not alone. BPH is the leading reason men visit a urologist.
You can measure your BPH symptoms by taking the International Prostate Symptom Score (IPSS) questionnaire. Sharing this information with your physician will help them understand the severity of your symptoms.
Even watchful waiting has its risks. Bladder outlet obstruction could result in irreversible bladder damage if left untreated.
BPH Treatment Options
If you have been diagnosed with an enlarged prostate due to BPH, consult with your physician to determine which treatment is right for you.
Treatment Options may include:
Watchful Waiting: Watchful waiting involves making behavioral changes such as decreasing your fluid intake prior to bedtime, regular physical activity, and avoiding caffeine and alcohol. For men with mild symptoms this may be a viable option until their condition shows signs of worsening. However, it is important to note that, in a study, 87% of men who elected watchful waiting experienced a worsening of symptoms over a 4-year time period.
Medications: Your doctor may prescribe BPH medication, just one or even a combination of BPH medications that may help alleviate your urinary symptoms. These medications are effective and may improve your BPH symptoms; however, some men may not experience enough relief. In addition, some men experience bothersome side effects from BPH medication. Either or both of these things may lead them to discontinue the medication.
Minimally Invasive Treatments: These are procedures which can often be done as an outpatient procedure with milder forms of anesthesia than what is used in a hospital operating room. They typically have fewer complications thantraditional surgery.
The UroLift® System uses small implants to open the prostate blockage and does not require heating or cutting tissue. The procedure is typically performed under local anesthesia in a doctor’s office and patients typically return home the same day without a catheter. The UroLift System offers rapid symptom relief with a low risk profile.
As the procedure does not destroy tissue, recovery on average is more rapid than for TURP patients. It is the only leading BPH procedure shown to provide symptom relief while also preserving sexual function. Patients may experience short-term pelvic pain and light blood in the urine.
*No instances of new, sustained erectile or ejaculatory dysfunction in the L.I.F.T. pivotal study. Another minimally invasive BPH treatment is Rezum™ Water Vapor Therapy. This is a form of thermal therapy in which the prostate is heated with steam. Another form of thermal therapy called Transurethral Microwave Therapy(TUMT) uses microwaves to destroy tissue. They can typically be done at a doctor’s office with local anesthesia and patients can usually go home the same day as the procedure. These procedures generally have fewer complications than traditional surgery. A catheter is typically required for a few days after the procedure and recovery may take a few weeks.
Traditional Surgery: Common traditional BPH surgeries include transurethral resection of the prostate (TURP) and laser photoselective vaporization of the prostate (GreenLight™ laser PVP). These procedures are the most aggressive treatment option and can be effective. Recovery can take time and is accompanied by a higher risk of complications. Less commonly performed surgeries include Aquablation® therapy and Holmium Laser Enucleation of the Prostate (HoLEP). Aquablation® therapy uses a high-pressure water jet to remove prostate tissue, whereas HoLEP uses a laser to remove the prostate tissue. Traditional surgery can offer significant relief of symptoms and help you improve quality of life.
The UroLift® System Treatment
What is the UroLift System?
The UroLift System is a proven minimally invasive procedure to treat an enlarged prostate. It is the only leading enlarged prostate procedure that does not require heating, cutting, or destruction of prostate tissue. The procedure is typically performed as a same-day outpatient procedure, including the office setting, under local anesthesia.
Advantages of the UroLift System
- Safe and effective
- Risk profile better than reported for surgical procedures such as TURP
- Rapid symptom relief, better than reported for medications
- Only leading BPH procedure shown not to cause new and lasting sexual dysfunction
- Covered by Medicare, national, and commercial plans
- Typically no catheter required after treatment
How Does The UroLift® System Work?
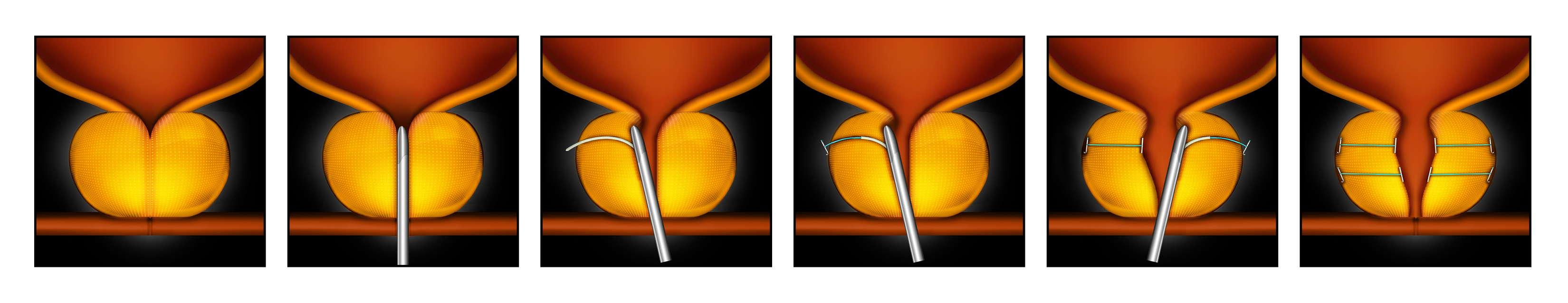
STEP 1
STEP 2
STEP 3
Step 1: The UroLift Delivery Device is placed through the obstructed urethra to access the enlarged prostate.
Step 2: Small UroLift Implants are permanently placed to lift and hold the enlarged prostate tissue out of the way and increase the opening of the urethra.
Step 3: The UroLift Delivery Device is removed, leaving an open urethra designed to provide symptom relief.
Indicated for the treatment of symptoms of an enlarged prostate up to 100 cc in men 45 years or older. As with any medical procedure, individual results may vary. Most common side effects are temporary and include pain or burning with urination, blood in the urine, pelvic pain, urgent need to urinate, and/or the inability to control the urge. Rare side effects, including bleeding and infection, may lead to a serious outcome and may require intervention. Speak with your doctor to determine if you may be a candidate.
Frequently Asked Questions
The UroLift Implants are small permanent implants that hold the obstructing prostatic lobes apart. They are deployed through a needle that comes out of the delivery device. Each implant is made with common implantable materials: nitinol, stainless steel, and suture. Typically, 4-6 implants are placed into the prostate.


